Positive outcome for ema's policy 0070, one year after the publication of the clinical trial results - Portolano Cavallo

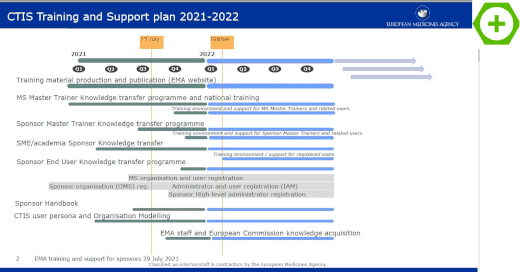
EMA's Clinical Trials Information System (CTIS) goes live – EJP RD – European Joint Programme on Rare Diseases
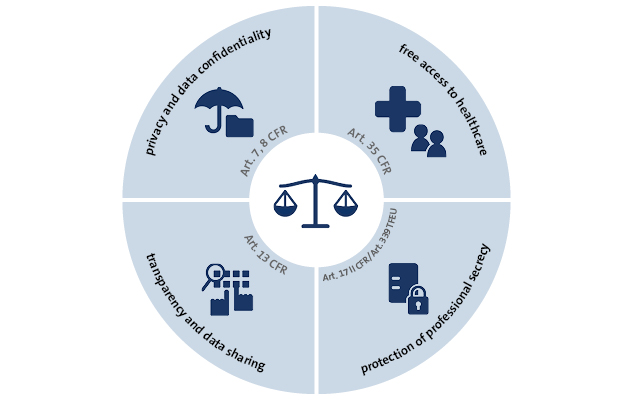
The obligatory sharing of clinical trial data in the European Union - datenschutz notizen | News-Blog der datenschutz nord Gruppe

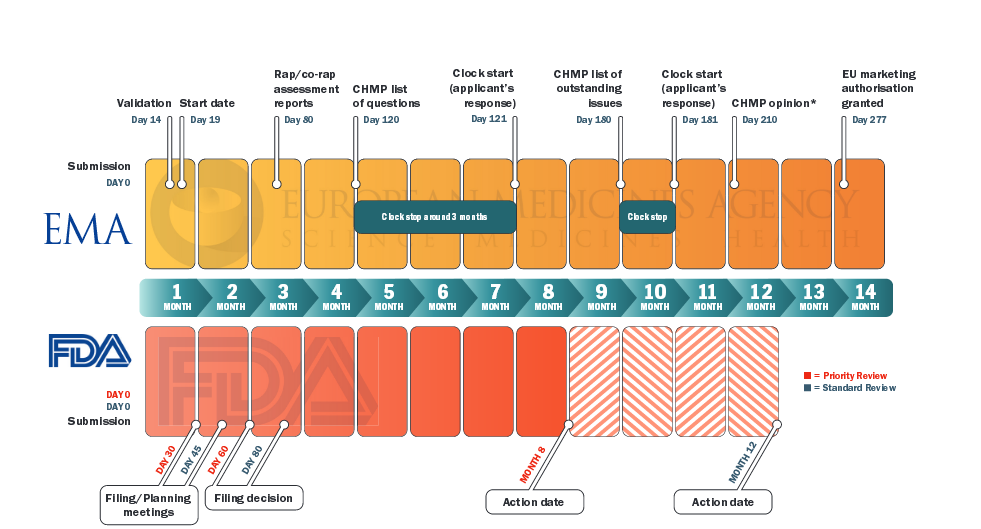
Europe - Clinical Trials Information System reaches major milestone towards go-live and application of the Clinical Trial Regulation - RIS.WORLD

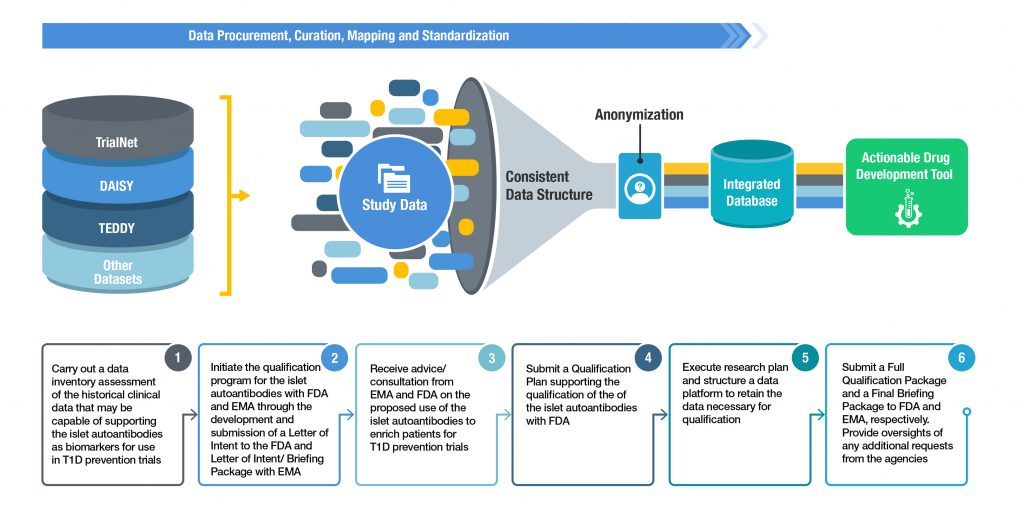
Call to pool research resources into large multi-centre, multi-arm clinical trials to generate sound evidence on COVID-19 treatments | European Medicines Agency

EMA: Points to consider on implications of COVID-19 on methodological aspects of ongoing clinical trials - Meditrial Helpline

Transparency of clinical trials and good governance should be included in the EMA extended mandate | European Alliance for Responsible R&D and Affordable Medicines

Clément Provansal on LinkedIn: Regulatory harmonisation of clinical trials in the EU: Clinical Trials