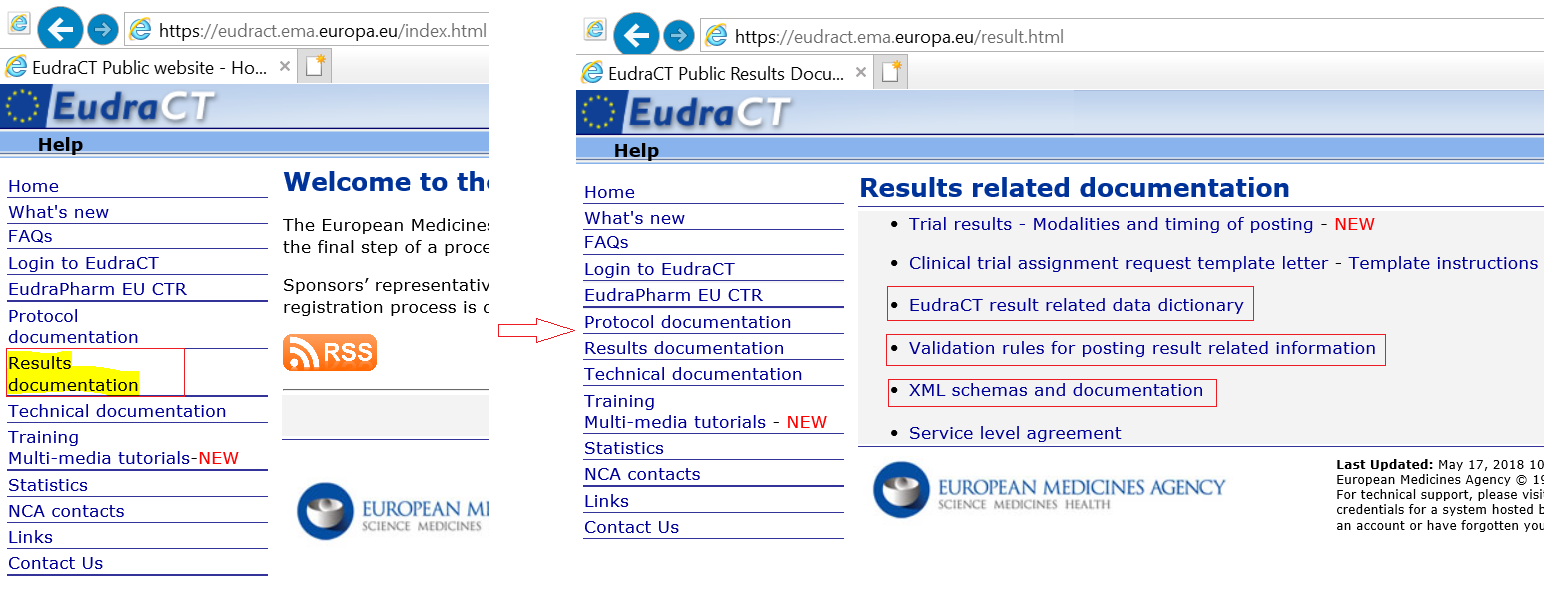
Compliance with requirement to report results on the EU Clinical Trials Register: cohort study and web resource | The BMJ

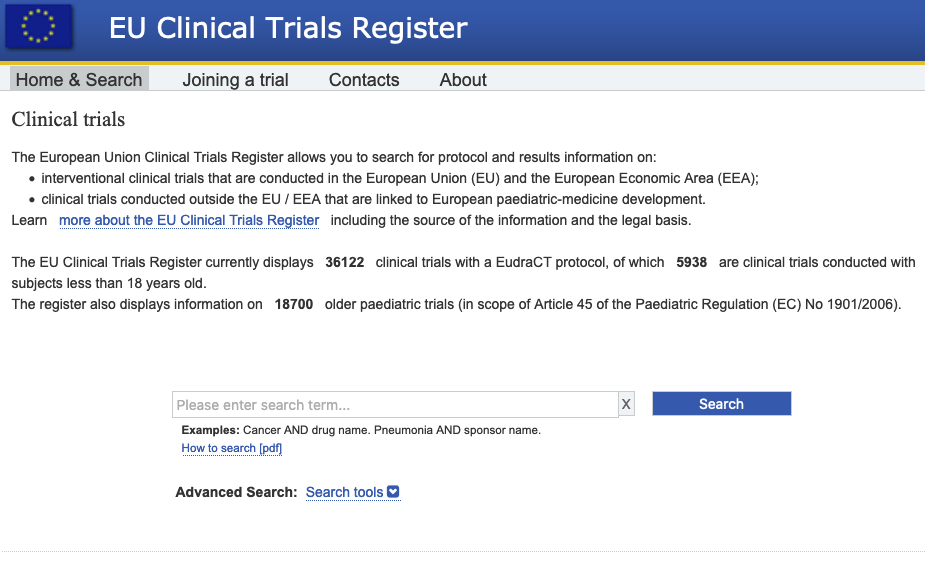
Flow of diagram of study. Abbreviation: eU-cTr, european Union clinical... | Download Scientific Diagram
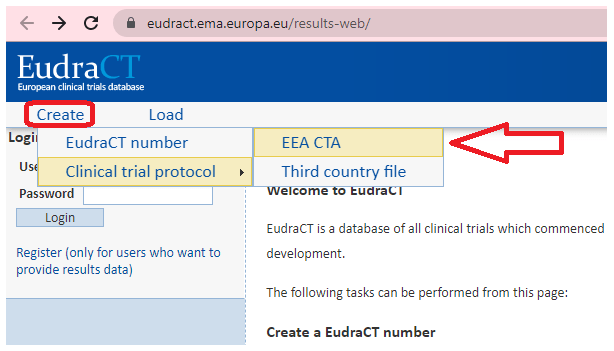
What Are the Documents Required for Clinical Trial Applications to Regulatory Authorities in Europe? - Sofpromed