Chinese Clinical Trial Register (ChiCTR) - The world health organization international clinical trials registered organization registered platform
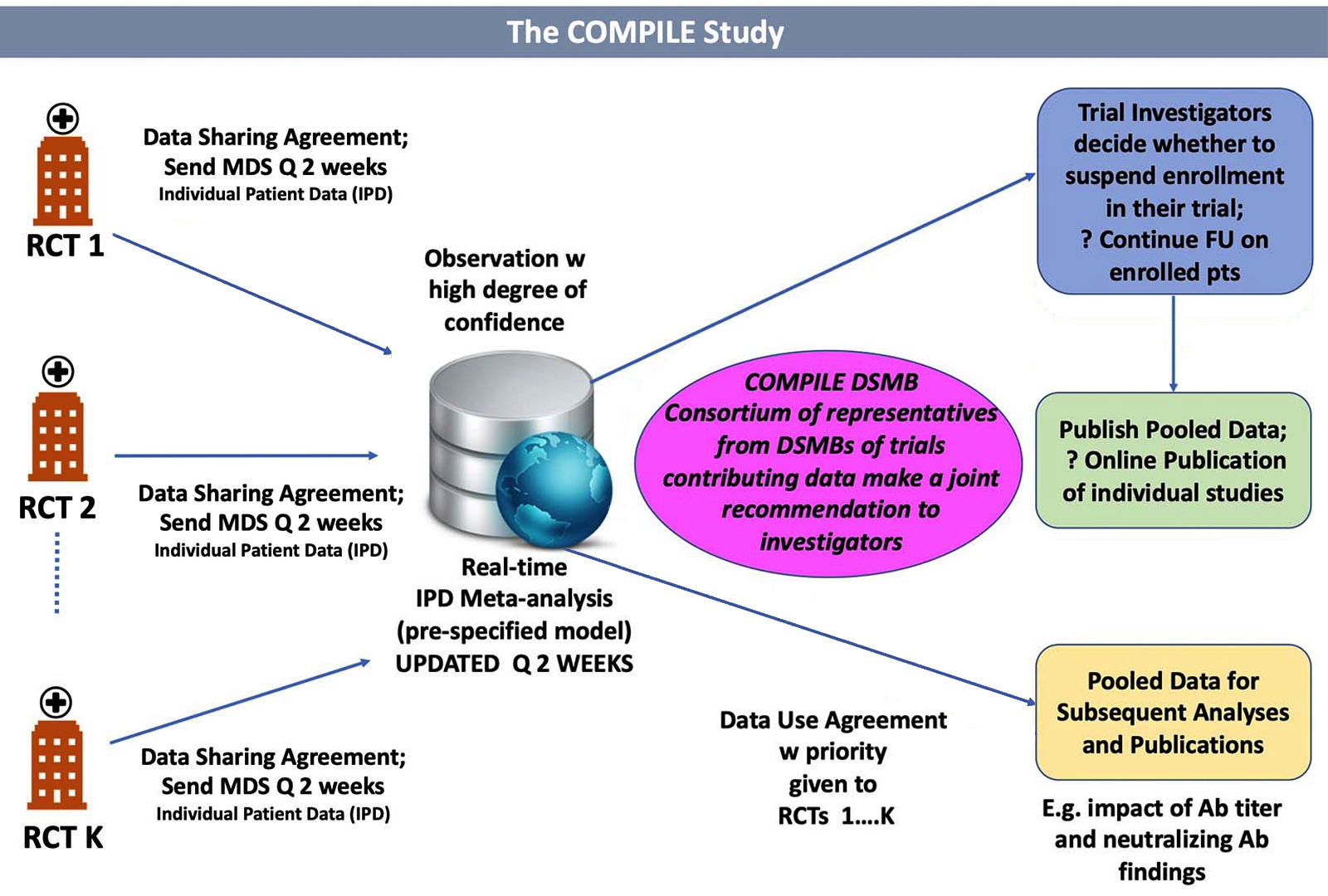
Continuous Monitoring of Pooled International Trials of Convalescent Plasma for COVID-19 Hospitalized Patients | NYU Langone Health
![PDF] Individual Participant Data (IPD) Meta-analyses of Randomised Controlled Trials: Guidance on Their Use | Semantic Scholar PDF] Individual Participant Data (IPD) Meta-analyses of Randomised Controlled Trials: Guidance on Their Use | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/1cff38f5e353e77f7e59bcdd131eb8cd986591d6/3-Table1-1.png)
PDF] Individual Participant Data (IPD) Meta-analyses of Randomised Controlled Trials: Guidance on Their Use | Semantic Scholar

How researchers should select the best outcomes for randomised clinical trials in paediatric dentistry? - Mendes - 2020 - International Journal of Paediatric Dentistry - Wiley Online Library
PLOS Medicine: Individual Participant Data (IPD) Meta-analyses of Randomised Controlled Trials: Guidance on Their Use

Direct and indirect comparison methods. IPD = individual patient data. | Download Scientific Diagram

PDF) Individual Participant Data (IPD) Meta-analyses of Randomised Controlled Trials: Guidance on Their Use

PDF) Sharing Individual Participant Data (IPD) within the Context of the Trial Reporting System (TRS)

Individual Patient-Level Data Sharing for Continuous Learning: A Strategy for Trial Data Sharing - National Academy of Medicine

Global Clinical Trial Data Sharing - The Multi-Regional Clinical Trials Center of Harvard and Brigham and Women's Hospital
Sharing Individual Participant Data (IPD) within the Context of the Trial Reporting System (TRS) | PLOS Medicine

Evaluation of Data Sharing After Implementation of the International Committee of Medical Journal Editors Data Sharing Statement Requirement. - Abstract - Europe PMC

Cochrane Workshop on Reviews Using IPD The Use of Individual Patient Data in Systematic Reviews Lesley, Stewart 1, Mike Clarke 2, Jayne Tierney 1 (Cochrane. - ppt download
PLOS Medicine: Sharing Individual Participant Data (IPD) within the Context of the Trial Reporting System (TRS)
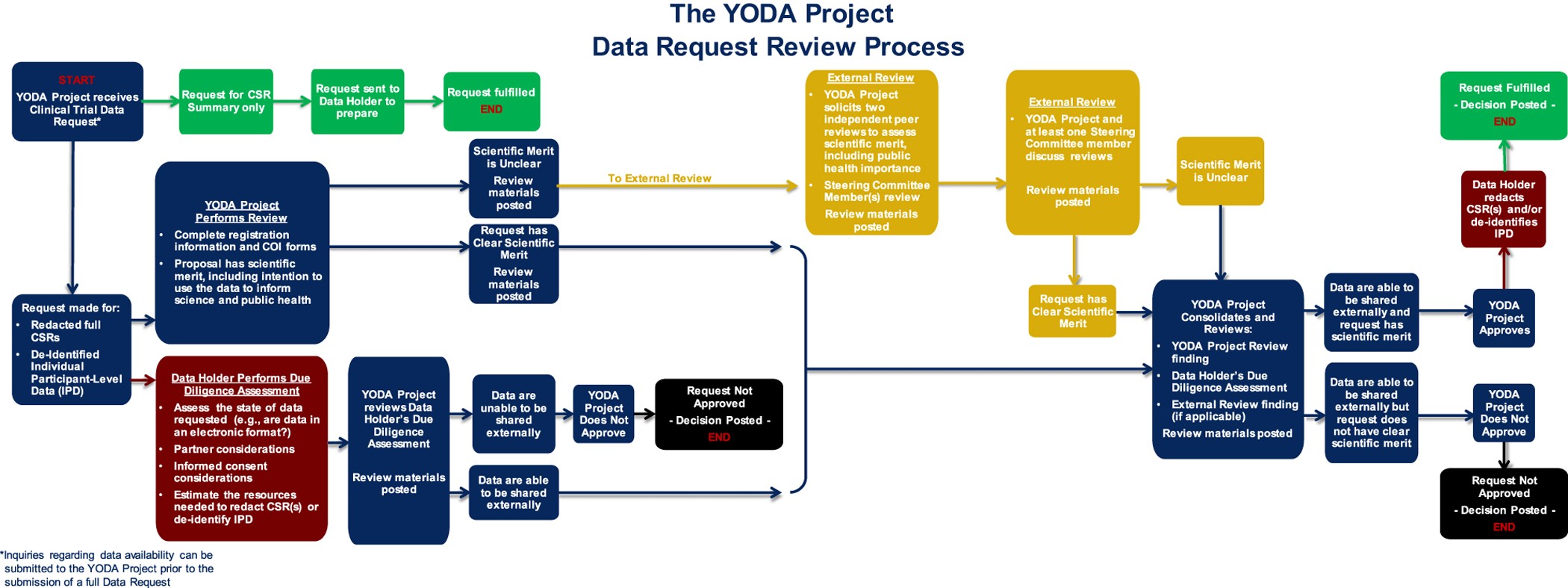
Overview and experience of the YODA Project with clinical trial data sharing after 5 years | Scientific Data
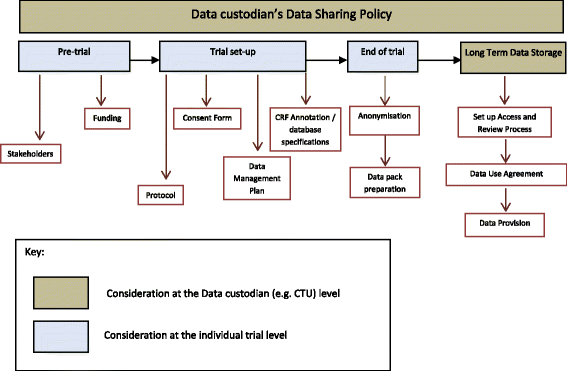
How should individual participant data (IPD) from publicly funded clinical trials be shared? | BMC Medicine | Full Text

PDF) Sharing Individual Participant Data from Clinical Trials: An Opinion Survey Regarding the Establishment of a Central Repository