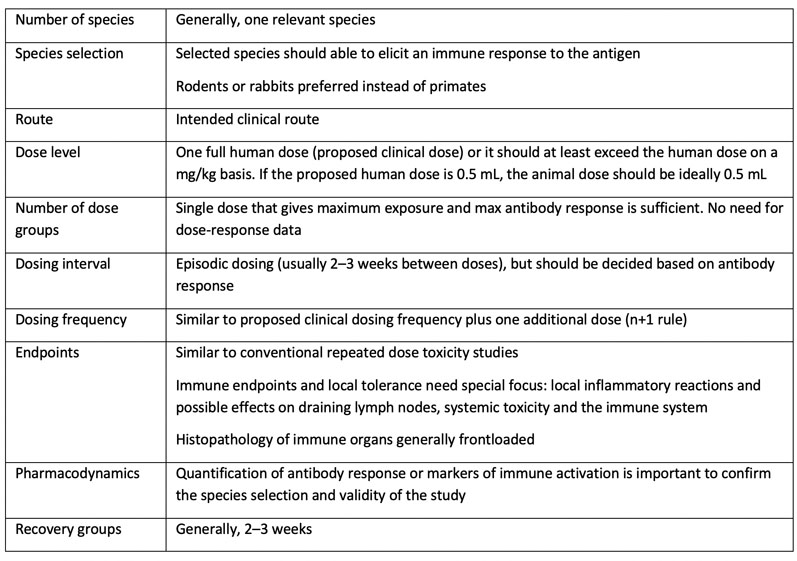
ICH M3 (R2): Non-Clinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceutica

Conclusions from non-clinical safety studies of the OPCs to be injected... | Download Scientific Diagram

ICH M3(R2) - Guidance on Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals

Table 1 from Non-clinical studies in the process of new drug development - Part II: Good laboratory practice, metabolism, pharmacokinetics, safety and dose translation to clinical studies | Semantic Scholar

InnoMed PredTox Consortium Presents Final Results from Non-clinical Safety Studies | Technology Networks
Non-clinical studies in the process of new drug Development--Part II: Good laboratory practice, metabolism, pharmacokinetics, safety and dose translation to clinical studies - Document - Gale Academic OneFile
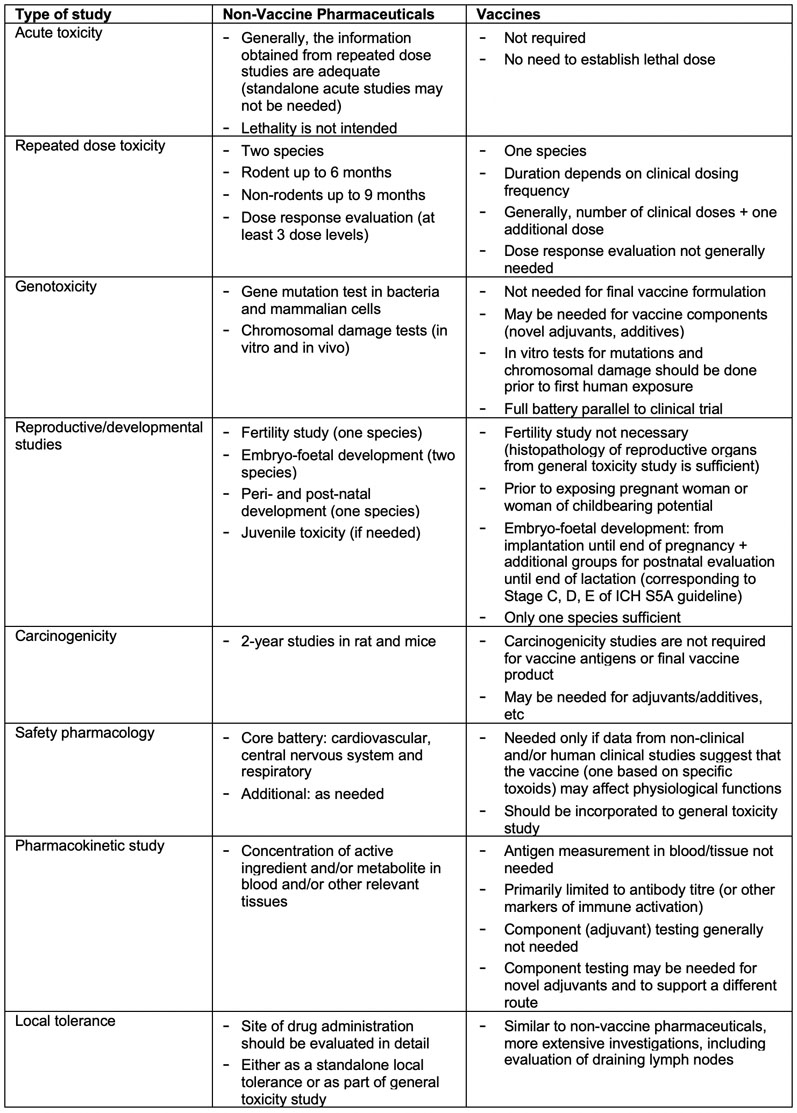
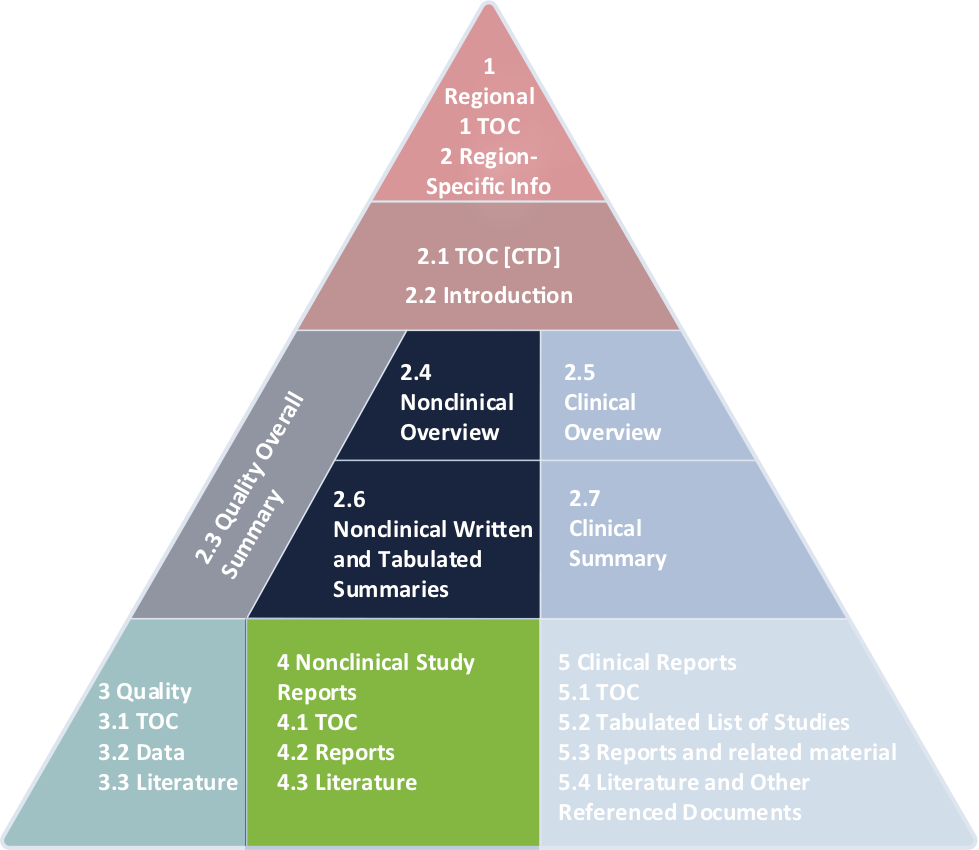




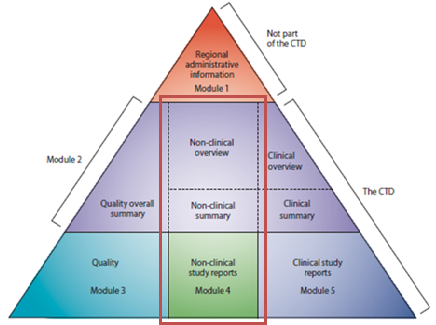


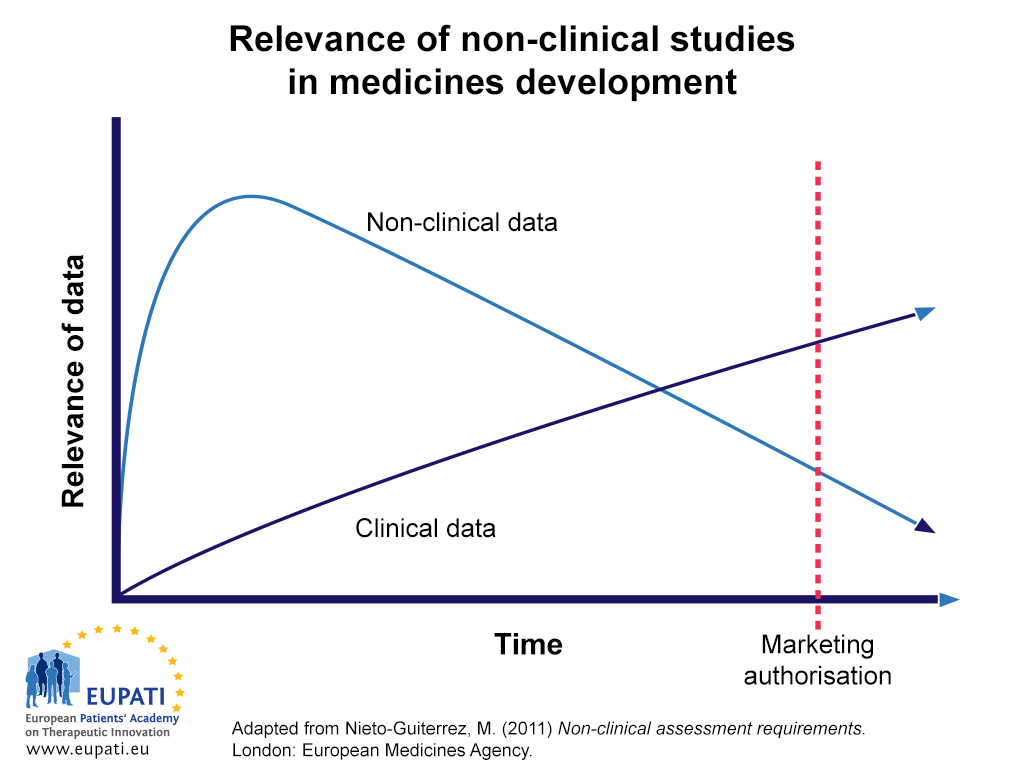
![PDF] An introduction to little-known aspects of nonclinical regulatory writing | Semantic Scholar PDF] An introduction to little-known aspects of nonclinical regulatory writing | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3c0319091233e268c650a8558c6a629ecf8655fe/5-Table1-1.png)